THE DIRECT FUNCTIONAL INTERRELATION OF SENSORY CORTEX AND OPTIC THALAMUS12 3 [21]
J.G. Dusser de Barenne and W.S. McCulloch
Introduction
The optic thalamus is the last subcortical relay-station for all corticopetal sensory4systems. From its lateral, ventro-lateral and ventral nuclei originate the thalamo-cortical neurons to terminate in the sensory cortex. However, these nuclei are connected with the sensory cortex not only through these corticopetal fibers but also through innumerable corticofugal neurons, beginning in the sensory cortex and ending in these very same thalamic nuclei.(1) These two-way connections indicate a close functional interrelation between thalamus and sensory cortex. This interrelation was proposed first in 1911 by Head,(2) who, to explain the spontaneous pains and hypersensitivity of the skin frequently a component of the “thalamic syndrome,” advanced the hypothesis that cortico-thalamic impulses inhibit thalamic activity. On the basis of his experiments with local strychninization of the monkey’s cortex Dusser de Barenne in 1924(3) advanced two hypotheses, 1. that such strychninization “sets on fire” a much larger portion of the sensory cortex than the area strychninized, 2. that under these conditions the corresponding thalamic nucleus also is “fired” (3). The first of these hypotheses has been verified and amplified recently.(4) In this paper we bring the verification of the second hypothesis.
Inasmuch as the local strychninization5 anywhere in the sensory cortex produces “strychnine-spikes” in the electrocorticogram (ECG) of functionally related areas,(4) it was plausible to expect that the same local strychninization of this cortex might also “fire,” i.e., produce strychinine-spikes in the electrothalamogram (ETG) of the related thalamic nucleus or nuclei. If this surmise proved to be correct, one would have here a physiological method for the investigation of the direct cortico-thalamic functional relations. To attack this problem it was necessary to know in which thalamic nucleus or nuclei the face, arms and legs are represented sensorially, i.e., to know the functional localization in the thalamus of the monkey. This localization was established in 1934-1935 by intrathalamic strychninization, in combination with “clinical” observation of the ensuing sensory disturbances.(5)
Once this functional localization was known the experiments to be reported in this paper were, in principle, very simple: concentric needle-electrodes were introduced into a particular nucleus of the thalamus and the ETG recorded before and after the local strychninization of the various subdivisions of the sensory cortex.
These experiments have established the following, direct functional relation of the sensory cortex to the thalamus: each thalamic nucleus is “fired” by the local strychninization of that, and only that, subdivision of the sensory cortex which subserves sensation of the same part of the body.
A second group of experiments in which the ECG of the various subdivisions of the sensory cortex was taken before and after intrathalamic strychninization, has shown the following direct functional relation of the sensory nuclei of the thalamus to the sensory cortex: the injection of strychnine into any particular sensory thalamic nucleus “fires” that, and only that, subdivision of the sensory cortex which subserves sensation in the same part of the body.
Method
All experiments were performed on Macaque monkeys (macaca mulatta), fully anaesthetized with Dial (Ciba)6; .45 cc. per kilogram body weight, part of the dose is given intraperitoneally, part of it intramuscularly. Then the animal was tied down upon the operating-table with its head fixed in a special headholder(5) so that the head was oriented in a definite, constant position in space. One sensori-motor cortex was then exposed, and with the micro-injection apparatus of Dusser de Barenne and Sager(6, 5) concentric needle-electrodes mounted on the barrel of a tuberculine-syringe, were introduced into the desired thalamic nucleus. The needle-electrodes consisted of a hypodermic needle (gauge no. 25), with its tip ground off conically, through which passed the central wire-electrode insulated to its tip. The central electrode protruded about 1½ mm. beyond the circumferential one. For recording the ETG the needle-electrodes were connected to a D.C. amplifier and cathode ray oscillograph. After taking the normal ETG the three subdivisions of the sensory cortex were locally strychninized, seriatim, ending with the subdivision functionally related to the thalamic nucleus into which the needle had been introduced. An ETG was taken after each strychninization. The experiment was repeated on the opposite hemisphere. Then the animal was killed by injection of 100 cc. of 15% formalin into each of the carotids. Half an hour later the brain was removed and placed in formalin; 15-20 hours later the brain was sectioned (in the frontal plane) in slices 2 mm. thick and thus the location of the needle in the thalamus identified. In the second group of experiments the ECGs were taken simultaneously with bipolar electrodes from the various subdivisions of the sensory cortex (A.C. amplifiers, 4-element Westinghouse oscil—lograph), before and after intrathalamic strychninization. This was performed as described by Dusser de Barenne and Sager.(5)
The nomenclature of the thalamic nuclei used here, as in the paper by Dusser de Barenne and Sager, is that of C. Vogt(7) and Friedemann.(8) This choice is purely for the practical reason that these authors have provided the most complete series of illustrations of the cercopithecus’ thalamus. There is no objection against this choice for the experiments on the macaque’s brain since Le Gros Clark and Boggon(9) have found that “the cytoarchitecture of the thalamus of Macaca corresponds extremely closely with that of Cercopithecus as described and figured by Friedemann (1911-12).” For the designation of the various cortical areas used here we refer to a previous paper.’
Results
We shall begin with two experiments on nucleus Vb, in which the face alone is represented sensorially.(5)
Protocol of an experiment with strychninization of the postcentral cortex.
November 18, 1936. Macacus. 2.82 kg. 9.00 a.m.-1.3 cc. Dial (⅔ of the dose intraperitoneally, ⅓ intramuscularly). Exposure of right hemisphere. Concentric needle-electrodes into thalamus by transverse route, intended to reach nucleus Vb (face-nucleus), inclination 35°, depth 16½mm. Central electrode to grid of amplifier, circumferential electrode (needle proper) to cathode.
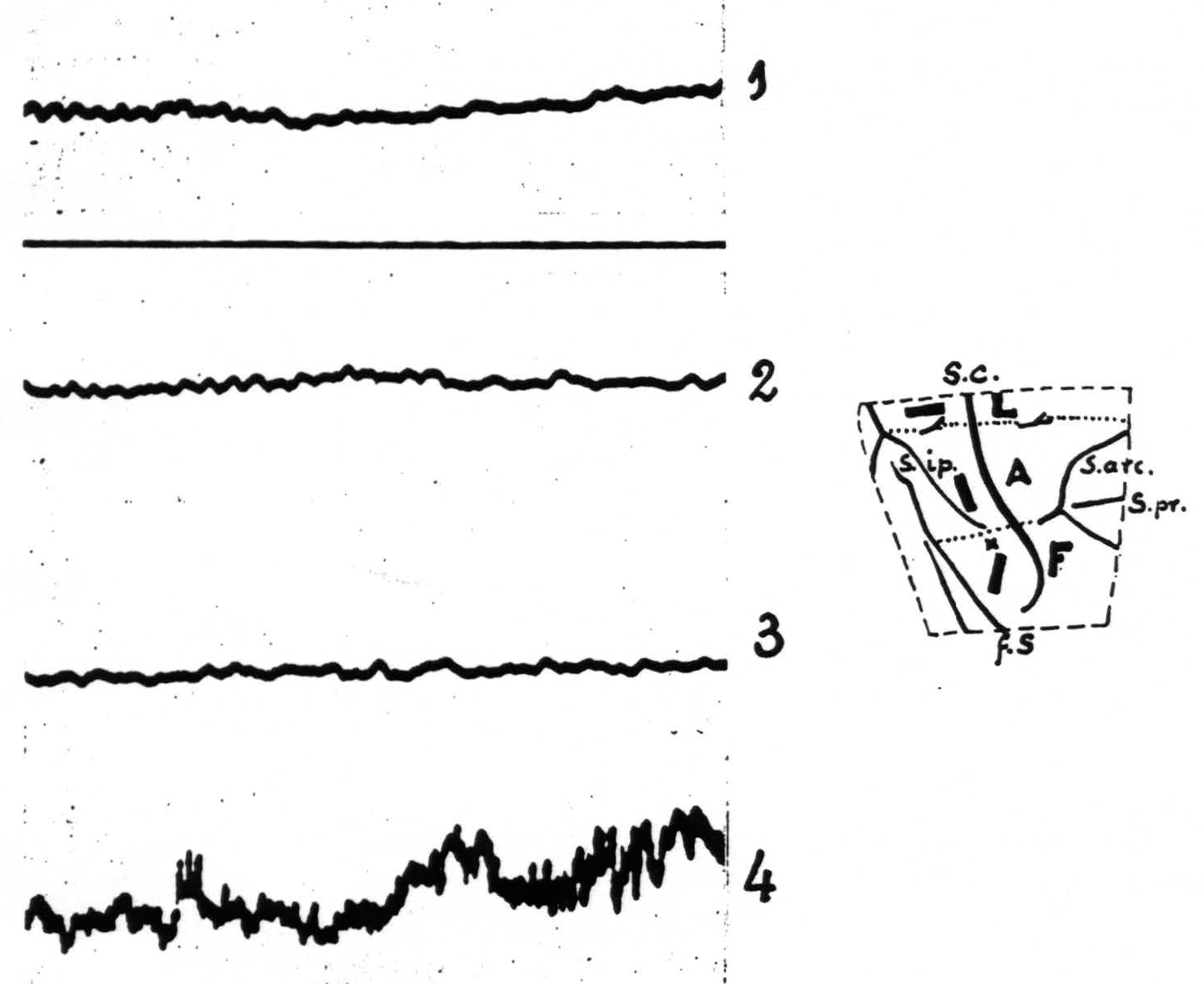
Figure 1. Experiment of November 18, 1936. Macaque monkey. Dial narcosis. Electrothalamogram (ETG) recorded from nucleus Vb (face-nucleus). Record 1——control. Record 2—10 minutes after strychninization of postcentral leg-area. Record 3—10 minutes after strychninization of postcentral arm-area. Record 4 —13 minutes after strychninization of postcentral face-area. Only in record 4 ETG shows “firing” of face-nucleus. Amplification in all records the same. Diagram indicates location of the 3 strychnine-applications and the site of entrance of the needle (x). The stippled lines indicate the boundaries between leg- and arm- (L and A) and arm- and face-subdivisions (A and F) of the exposed portion of the sensory cortex. Broken line indicates edge of opening in dura. S.c. =sulcus centralis; s.ip. =sulcus intraparietalis; f.S. =fissura Sylvii, s.arc. =sulcus arcuatus; s. pr. =sulcus principalis.
D.C. amplifier in combination with cathode ray oscillograph. Cambridge moving paper camera.
| 10.55. | Record 1 (Fig. 1) taken with lowest amplification. |
| 10.57. | Strychninization of postcentral *leg*-area by applying across its width an oblong piece of filter paper, moist with 3 per cent strychine-solution (dimensions of filter paper 1×3 mm.). |
| 11.07. | Record 2 (Fig. 1) taken. |
| 11.11. | Strychninization of *postcentral* arm-area by application of a similar strip of filter paper to lower half of postcentral gyrus, parallel to central sulcus. |
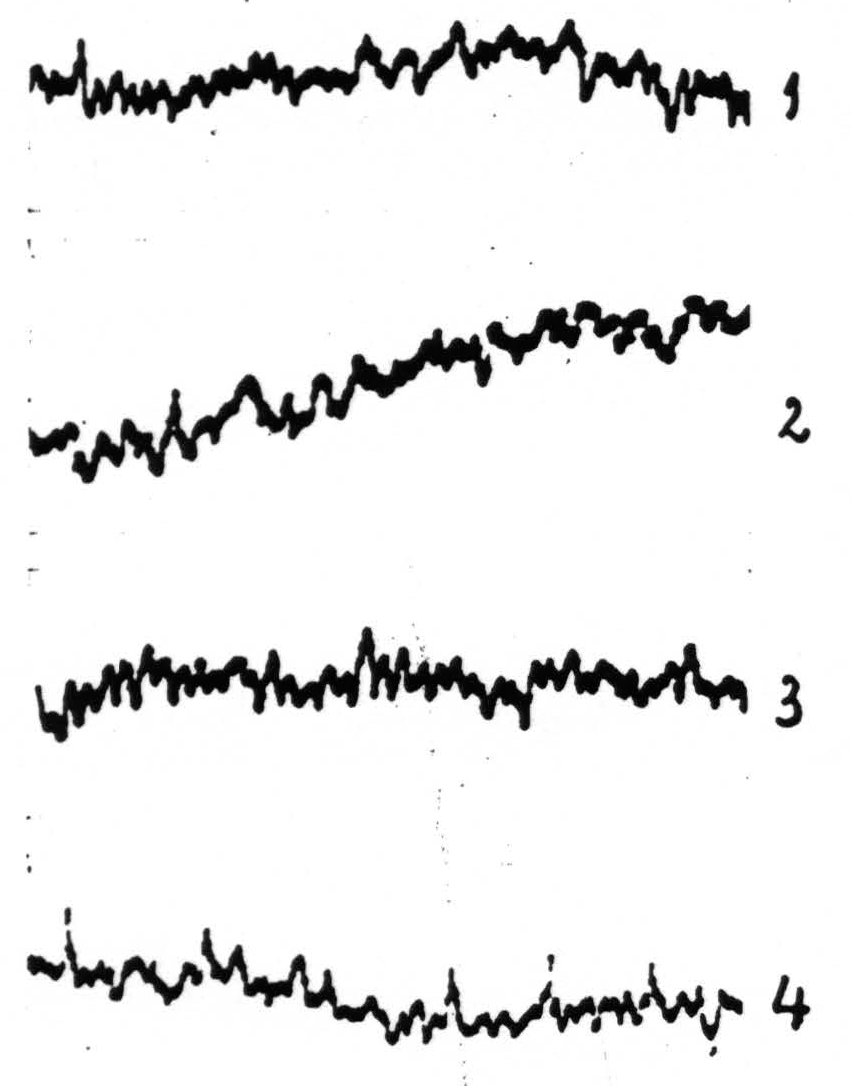
Figure 2. Experiment of November 23, 1936. ETG of nucleus Vb₂ (face-nucleus) of thalamus of macaca mulatta (Dial-narcosis) before (record 1) and after (records 2, 3 and 4) local strychninization of precentral sensory cortex, leg-, arm- and face-areas respectively. No apparent change in ETG of records 2 and 3 compared with that of record 1; definite “firing” in ETG in record 4, i.e., 11 minutes after local strychninization of pre-central face cortex.
| 11.21. | Record 2 (Fig. 1) taken. |
| 11.23. | Strychnine applied to |
| 11.36. | Record 4 (Fig. 1) taken. |
Autopsy: end of needle-track in nucleus Vb at level of Vogt’s plate no. 5 (7, 5).
In the following experiment the strychninizations were performed on the precentral sensory cortex.
November 23, 1936. Macaca mulatta. 2.80 kg.
| 9.15 a.m. | 1.3 cc. Dial (⅔ of dose intraperitoneally, ⅓ intramuscularly). |
| 11.30. | Animal not fully under. .1 cc Dial intramuscularly. |
| 2.00 p.m. | Animal completely anaesthetized. |
| 2.16. | Exposure of |
| 2.19. | Record 1 (Fig. 2) taken (amplification 3). |
| 2.21. | Strychninization of |
| 2.32. | Record 2 (Fig. 2) taken (same amplification as before). |
| 2.33½. | Strychninization of |
| 2.40. | Record 3 (Fig. 2 ) taken (same amplification). |
| 3.09. | Strychninization of |
| 3.20. | Record 4 (Fig. 2) taken (same amplification as before.) |
November 24. Autopsy: needle-track ends in plane between C. Vogt’s plates 5 and 6 in nucleus Vb₂.
From these two and similar experiments it follows (1) that the local strychninization of the face-subdivision of the sensory cortex results in typical changes in the ETG of the face-nucleus (Vb); (2) that the local strych-ninization of any portion of the two other subdivisions of the sensory cortex does not produce any changes in the ETG of this nucleus; (3) that this nucleus is “fired” by the local strychninization of the precentral as well as of the postcentral portion of the face-subdivision of the sensory cortex.
The same obtains for the arm- and leg-nuclei, i.e., that each is “fired” only by the local strychninization of the pre- or postcentral portion of the subdivision of the sensory cortex corresponding to it.
There is one point which needs further elucidation, namely the apparent contradiction which was mentioned in our paper on “functional organization in the sensory cortex of the monkey” (4, p. 83). There it was reported that strychnine-spikes appear in the ECG of both the leg- and arm-subdivisions of the sensory cortex upon local strychninization of either L.6a or A.6a. Those observations have shown that so far as this distribution of the strychnine-spikes in the cortex is concerned no functional boundary exhibits itself between L.6a and A.6a. However, the contrast between this absence of the functional boundary, so far as the ECG in concerned, and its presence in regard to the symptoms of sensory excitation upon local strychninization of these same areas is not a contradiction, for the ECG expresses the activity of the cortex at that level of the CNS, whereas the symptoms of sensory excitation depend upon lower levels, notably the sensory nuclei of the optic thalamus, upon which converge all sensory impulses from the periphery of the body. At present we know that local strychninization of the sensory cortex “fires” the sensory nuclei of the thalamus, and in this respect one finds a definite functional boundary between L.6a and A.6a, for the leg-nuclei are “fired” by local strychninization of L.6a, but not by that of A.6a, whereas the arm-nuclei are “fired” by local strychninization of A.6a, but not by that of L.6a.
The following protocol serves as an illustration.
November 30, 1936. Macaca mulatta. 3.38 kg.
| 9.10. | 1.52 cc. Dial (⅔ of dose intraperitoneally, ⅓ intramuscularly). |
| 10.00. | Animal completely under. |
| 10.02. | Exposure of |
| 10.05. | Concentric needle-electrodes into thalamus via transversal route. Intention to reach the dorsal portion of one of the arm-nuclei, IbIV or lbI (C. Vogt), inclination 33°, depth 19 mm. |
| 10.25. | Record 1 (Fig. 3) taken (control) and then a series of records from various areas of the leg- and arm-subdivisions of sensory cortex. |
| 10.37. | Local strychninization (1×3 mm.) of |
| 10.44. | Record 2 (Fig. 3) taken with same amplification as in record 1. Subsequently a series of records from various areas of sensory cortex was taken (L.6a, L.4, L.2, A.6a, A.4, A.2, L.6a).6 Needle left in thalamus until afternoon. |
| 2.31. | Record 3 (Fig. 3) taken as a control. |
| 2.40½. | Local strychninization of |
| 2.45. | Record 4 (Fig. 3) taken, followed by recording of ECG of various areas of the leg- and arm-subdivisions.2 |
December 1.
Autopsy: End of needle track in dorsal portion of nucleus IbI (Vogt’s plate 6 (arm nucleus)). Conclusion: local strychninization of L.6a did not “fire” this nucleus, whereas local strychninization of A.6a promptly “fired” this nucleus. Following the strychninization of L.6a (morning) and that of A.6a (afternoon) both entire leg- and arm-subdivisions were “fired.”

Figure 3. Experiment of November 30, 1936. ETGs of nucleus IbI (one of the arm-nuclei) of macaque’s thalamus. Dial-narcosis. Record 1 taken before, record 2 after local strychninization of L.6a. No change in ETG. Record 3 taken before, record 4 after local strychninization of A.6a. Prompt “firing” of nucleus lbl’. The entire leg- and arm-subdivisions were fired by each of the two strychninizations.
The second group of experiments mentioned above rounds out the picture of the direct functional interrelation of sensory cortex and thalamus. In these experiments intrathalamic strychnine-injections were performed, using the micro-injection technique described previously,2,3 and recording the ECG of various areas of the three subdivisions of the sensory cortex. The essential result in this group was that the injection of strychnine into any particular sensory nucleus of the thalamus “fired” that, and only that, subdivision of the sensory cortex, both precentral and postcentral, which subserves sensation in the same part of the body. This result must be interpreted as showing that in each particular nucleus of the thalamus, let us say, in each arm-nucleus originate thalamo-cortical neurons to terminate in the various areas of the arm-subdivision, and only of the arm-subdivision, of the sensory cortex; thus the strychninization of the perikarya of these neurons “sets on fire” the various areas of the arm-subdivision of the sensory cortex. The same relation obtains between the leg- and face-nuclei of the thalamus and the leg-and face-subdivisions of the sensory cortex respectively.
Finally it must be stated that (1) local strychninization of white matter (e.g., corona radiata, internal capsule, corpus callosum) in our experience never “fires” grey matter; (2) local strychninization of masses of grey matter having no direct corticipetal connections with the sensory cortex (e.g., nucleus caudatus, medial nuclei of the thalamus) never “fires” this region of the cortex.
Discussion
Apart from the results, reported above, to which we shall return below, these experiments provide a check upon the extent, location and subdivision of the sensory cortex on the outer surface of the macaque’s brain,’ and upon the functional identification of the macaque’s sensory nuclei in the thalamus.’ In this connection two crucial findings should be reported here: 1. local strychninization without the sensory cortex never “fired” the face-, arm- or leg-nuclei of the thalamus; 2. local strychninization anywhere within the sensory cortex never “fired” the medial nuclei of the thalamus (mag, mapt, mapl, maga, magp, mα).
Together, all the results of the present investigation are in entire harmony with the results of the previous work and complete the confirmation of the two hypotheses advanced in 1924(3) to explain how strychninization of only a few square millimeters of the sensory cortex induces symptoms of sensory excitation in a large portion of the body. That local strychninization of grey matter in the CNS with recording of the electrical activity of grey matter in the CNS reveals directed functional relations has been shown in a previous paper.’ Therefore, the only construction which can be put on the facts here established by physiological methods is that there exist between the sensory cortex and the sensory nuclei of the thalamus directed functional relations: 1. from each subdivision of the sensory cortex to, and only to, the corresponding sensory thalamic nucleus or nuclei, 2. from each sensory thalamic nucleus to, and only to, the corresponding subdivision of the sensory cortex. This is schematized in Fig. 4.
The three principal features in this diagram are:
- the essential similarity of the three sensory systems for face, arms and legs;
- that each system after reaching the cortex feeds back into the thalamus;
- that throughout the entire course of these three systems functional boundaries are maintained.
The diagram shows these boundaries only for one frontal plane, but it must be pointed out that they are present throughout the entire fronto-occipital extent, both in the thalamus and in the cortex. Thus the boundary indicated in the diagram between the leg- and arm-subdivisions of the sensory cortex extends all the way from the interparietal sulcus to the anterior margin of area 6a, as represented in the diagrams of the extent, location and subdivision of the sensory cortex previously given."
The apparent contradiction mentioned on p. 180 leads to an important point. Two observations are relevant here:
- So far as the cortical distribution of the strychnine-spikes in the leg-and arm-subdivisions upon local strychninization of L.6a or A.6a is concerned, no functional boundary between the two subdivisions exhibits itself.
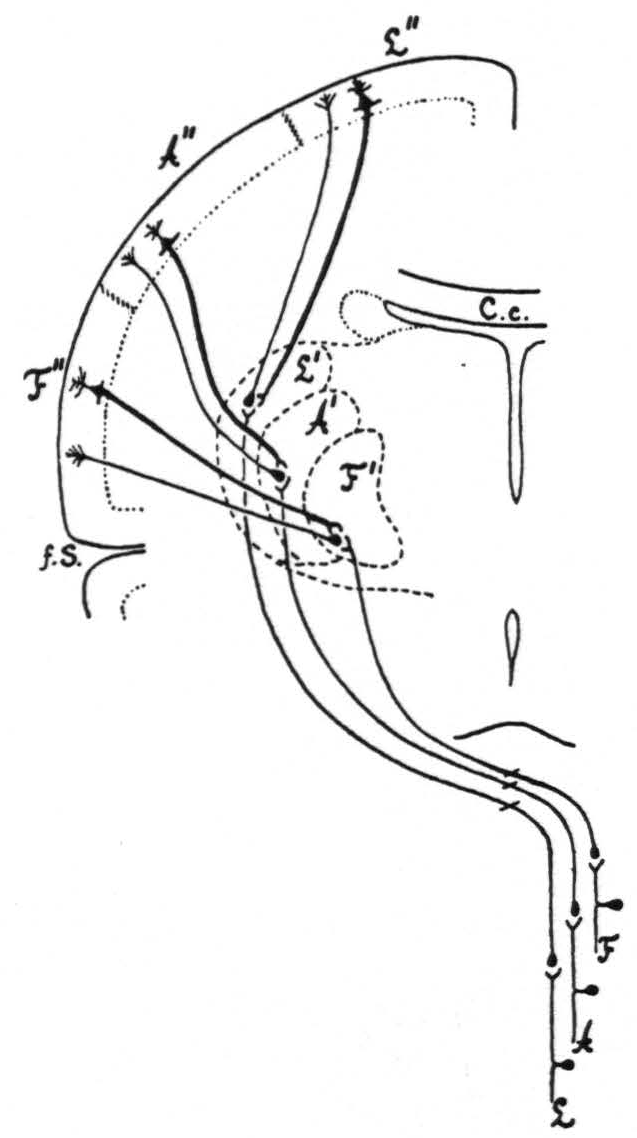
Figure 4. Diagrammatic representation of the organization of the three main sensory systems (for the legs, arms and face). All internuncial neurons between the subthalamic sensory neurons and the thalamo-cortical neurons have been omitted, to simplify as much as possible the diagram.
- Notwithstanding the fact that both entire leg- and arm-subdivisions of the sensory cortex are “fired” by local strychninization of either L.6a or A.6a, an animal, when “clinically” observed (without narcosis), discretely refers his symptoms of sensory excitation (paraesthesiae, hyperaesthesia and hyperalgesia) to the legs, and only to the legs, after local strychninization of L.6a, and conversely to the arms, and only to the arms, after local strychninization of A.6a.
These observations throw new light on the observation of Dusser de Ba-renne and Sager that the symptomatology after local intrathalamic strychninization in the acutely and totally decorticated cat was indistinguishable from the symptomatology in the animal with cortex intact. From that observation the authors concluded that in the case of strychninization of the thalamus “the sensory cortex plays no role in the elaboration of the ensuing sensory disturbances” (2, p. 246).
In this respect the cortex is (in the cat) not only unnecessary but is even unable (in the monkey!) to distort the distribution of the sensory disturbances determined by the hyperactive thalamic nucleus or nuclei. The functional boundaries remain. For the “firing” of the two entire leg- and arm-subdivisions of the sensory cortex by local strychninization within either L.6a or A.6a does not deter the animal from projecting his sensory disturbances discretely upon that part of the body subserved sensorially by the particular thalamic structure “fired” by the feed-back from the strychninized cortical area.
It follows that the part of the body to which the animal refers his symptoms of sensory excitation is determined not by the subdivision or subdivisions of the sensory cortex “fired,” but by the thalamic nucleus or nuclei “fired” by the direct feed-back from the strychninized area of the sensory cortex.
Thus, though both hypotheses advanced by Dusser de Barenne in explanation of his observations in 1924 have been substantiated, the second hypothesis, namely that local strychninization of any subdivision of the sensory cortex “sets on fire” the corresponding thalamic nucleus or nuclei, is evidently the crucial one. For all these observations and considerations imply not only the predominance of the thalamus in sensation, but that this last subcortical sensory station determines the reference of sensation by the individual, even contrary to coexisting cortical activity.
We must now discuss briefly the hypotheses of Head and Holmes(2) concerning the functional interrelation of the thalamus and cortex in the production of sensation.
In their well-known diagram(2) they represent an internuncial neuron from the ventro-lateral region of the thalamus to the medial nuclei, which they regard as the “essential center” of the thalamus. The cortico-thalamic neurons ending in the ventro-lateral region of the thalamus exert, according to Head and Holmes, an inhibitory influence upon this region of the thalamus. They assume that with interruption of these cortico-thalamic fibers the activity of the thalamus is released from its normal cortical restraint, and that the afferent impulses, reaching the ventro-lateral region of the thalamus, flow over uncontrolled to the medial thalamic region, which “is the centre of consciousness for certain elements of sensation” (l.c. p. 181), “mainly occupied with the affective side of sensation” (2).
This attractive conception, however, is open to criticism. Although in the cat the medial nuclei are endowed in some way with sensory functions’, this is not so in the monkey’. Apparently in this respect evolution of thalamic structures and functions has resulted in a definite shift. It is highly improbable that in the evolution of the human brain phylogeny should have reversed itself, backward toward the stage of the cat instead of onward from the stage of the monkey. In the second place the inhibitory function of the cortico-thalamic pathways is by no means established; in fact it was and is purely hypothetical. In all experiments presented here the strychninization of a number of the cells of origin of these cortico-thalamic neurons in the sensory cortex resulted in a “firing” of the corresponding thalamic nuclei, just as, in the earlier experiments of Dusser de Barenne’, it resulted in symptoms of sensory excitation. These are results which are the reverse of what could be expected on the basis of the hypotheses of Head and Holmes. Yet there may be some truth in them, for we have found that the local strychninization of two particular regions, areas 4-s and 1, of the monkey’s sensory cortex results in a temporary suppression of the electrical activity of area 44. However, this suppression in the case of area 4-s does not depend upon the direct cortico-thalamic connections which Head and Holmes had in mind and with which the present paper dealt. This suppression involves indirect cortico-thalamic relations and must be taken up in a subsequent paper.
Summary
The present paper deals with two groups of experiments:
- the effect of local strychninization of the sensory cortex upon the electrical activity of the optic thalamus, i.e., upon the electrothalamogram (ETG),
- the effect of local strychninization of the sensory thalamic nuclei upon the electrical activity of the sensory cortex, i.e. upon its electrocorticogram (ECG).
In the first group with local strychninization of the cortex it was found that each thalamic nucleus is “fired” from that, and only that, subdivision of the sensory cortex, subserving sensation in the same part of the body. In the second group of experiments it was found that local strychninization of any particular sensory thalamic nucleus “fires” that, and only that, subdivision of the sensory cortex which subserves sensation in the same part of the body.
Sharp functional boundaries exhibit themselves between the adjoining sensory systems.
The discussion indicates the significance of these observations in regard to the dominance of the thalamus over the sensory cortex in sensory disturbances, even when initiated by excitation from the sensory cortex.
Footnotes
References
Monakow, C. von. Experimentelle und pathologisch-anatomische Untersuchungen über die Haubenregion, den Sehhügel und die Regio subthalamica. Arch. Psychiat., 1895, 27: 1-128, 386-478.
Head, H., and Holmes, G. Sensory disturbances from cerebral lesions. Brain, 1912, 34: 102-254.
Dusser de Barenne, J. G. Experimental researches on sensory localization in the cerebral cortex of the monkey (Macacus). Proc. roy. Soc., 1924, B 96: 271-291. See also Dtsch. Z. Nervenheilk., 1924, 83: 273-299.
Dusser de Barenne, J. G., and McCulloch, W. S. Functional organization in the sensory cortex of the monkey (Macaca mulatta). J. Neurophysiol., 1938, 1: 69-85.
Dusser de Barenne, J. G., and Sager, O. Sensory functions of the optic thalamus of the monkey (Macacus rhesus). Symptomatology and functional localization investigated with the method of local strychninization. Arch. Neurol. Psychiat., Chicago, 1937, 38: 913-926.
Dusser de Barenne, J. G., and Sager, O. Über die sensiblen Funktionen des Thalamus opticus der Katze. Untersucht mit der Methode der örtlichen Strychninvergiftung; allgemeine Symptomatologie and funtionelle Lokalisation. Z. ges. Neurol. Psychiat. 1931, 133: 231-272.
Vogt, C. La myéloarchitecture du thalamus du cercopithèque. J. Psychol. Neurol., Lpz., 1909, 12 (Erg. Heft): 295-324.
Friedemann, M. Die Cytoarchitektonik des Zwischenhirns der Cercopitheken mit besonderer Berücksichtigung des Thalamus opticus. J. Psychol. Neurol., Lpz., 1911, 18 (Erg. Heft 2): 309-378.
Le Gros Clark, W. E., and Boggon, R. H. The thalamic connections of the parietal and frontal lobes of the brain in the monkey. Philos. Trans., 1935, B, 224: 313-359.
For further research:
Wordcloud: Activity, Animal, Areas, Arm, Arm-Subdivisions, Barenne, Body, Boundary, Connections, Cortex, Direct, Dusser, Ecg, Entire, Experiments, Face, Fig, Figure, Fired, Following, Functional, Head, Leg, Local, Mm, Monkey, Neurons, Nuclei, Nucleus, Observations, Paper, Portion, Postcentral, Precentral, Present, Record, Region, Relations, Results, Sensation, Sensory, Strychninization, Subdivision, Taken, Thalamic, Thalamus, Various, Vb
Keywords: Cortex, Nuclei, Monkey, Inquiry, Thalamus, Experiments, Connections, Strychninization, Neurons
Google Books: http://asclinks.live/a5tx
Google Scholar: http://asclinks.live/rpi6
Jstor: http://asclinks.live/87g1